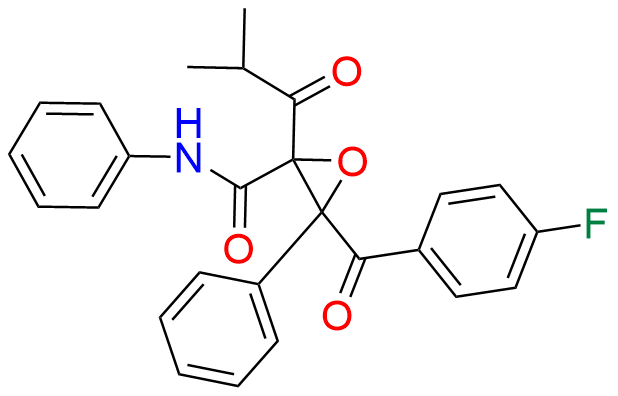
Atorvastatin EP Impurity D
| CAT. No. | CP-A32004 |
|---|---|
| CAS. No. | 148146-51-4 |
| Mol. F. | C26H22FNO4 |
| Mol. Wt. | 431.46 |
| Stock Status | In Stock |
Product Description
- Category:Impurity Standards
- Synonyms:Atorvastatin USP Related Compound D ; Atorvastatin Diketo Epoxide ; Atorvastatin Degradation Product - ATV-FXA1
- Chemical Name:3-(4-Fluorobenzoyl)-2-(2-methyl-1-oxopropyl)-N,3-diphenyloxiranecarboxamide