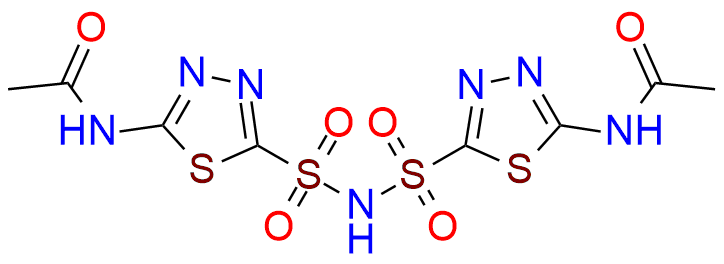
Acetazolamide EP Impurity F
| CAT. No. | CP-A6006 |
|---|---|
| CAS. No. | 80495-47-2 |
| Mol. F. | C8H9N7O6S4 |
| Mol. Wt. | 427.44 |
| Stock Status | In Stock |
Product Description
- Category:Impurity Standards
- Synonyms:Acetazolamide dimer (USP)
- Chemical Name:N-[5-[(5-Acetamido-1,3,4-thiadiazol-2-yl)sulfonyl]sulfamoyl-1,3,4-thiadiazol-2-yl]acetamide (as per EP)