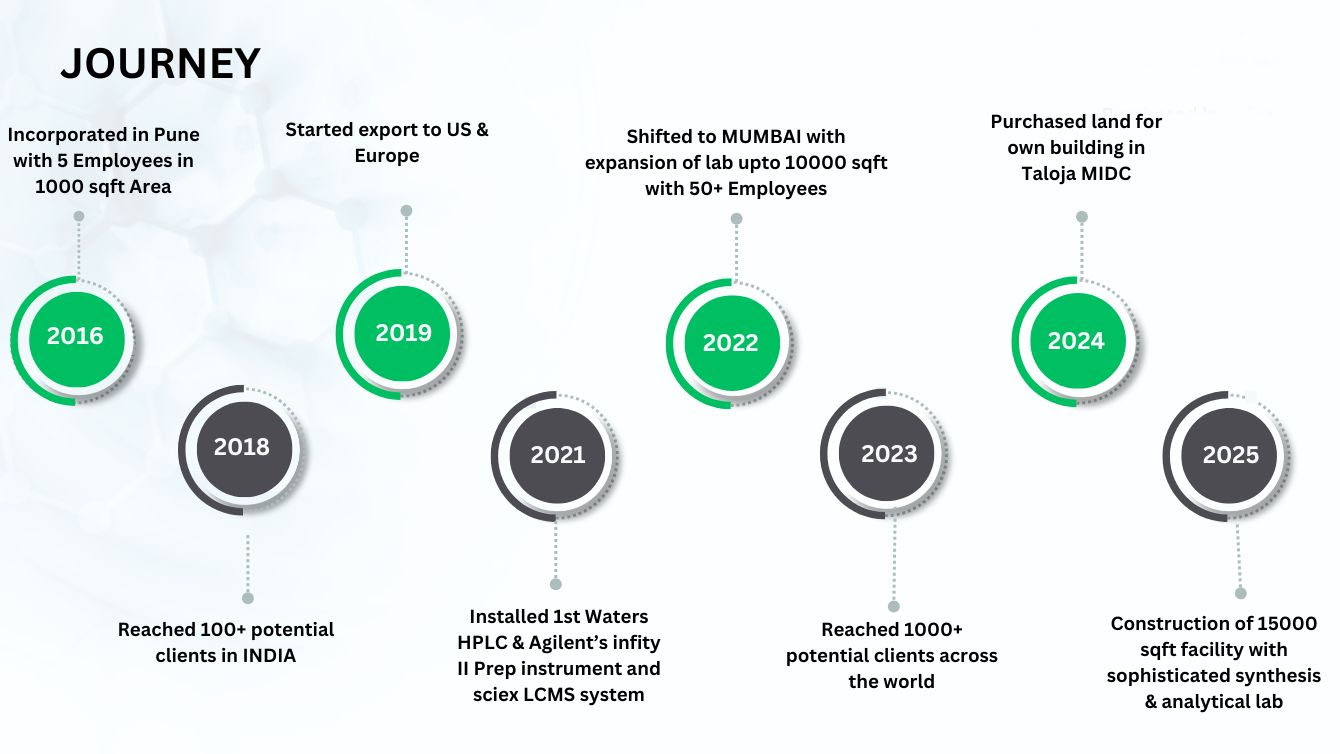
Chemicea Pharmaceuticals Pvt. Ltd., founded in 2016 and based in Navi Mumbai, is a leading manufacturer and global exporter of pharmaceutical impurity reference standards. The company specializes in the synthesis of process impurities, degradation products, and nitrosamine impurities, supporting pharmaceutical quality and regulatory compliance worldwide. Co-founded by Mr. Deepak Pawar and Mr. Santosh Chindhe both M.Sc. Organic Chemistry graduates from the University of Pune. Chemicea is widely recognized for its scientific innovation, stringent quality standards, and customer-centric approach. In addition to its technical expertise, Chemicea is committed to academic collaboration and environmental responsibility through initiatives such as research partnerships and community-focused programs.

At Chemicea, we synthesize and supply high-purity pharmaceutical impurity reference standards—including nitrosamines (NDSRIs and related metabolites), glucuronides, API adducts, building blocks and stable-isotope-labeled compounds. Our catalogue exceeds 7,000 items, with 300+ nitrosamines in ready stock, enabling rapid support for impurity profiling, method validation, drug development and regulatory submissions worldwide.
Beyond reference standards, our synthetic chemistry services cover small molecules synthesis, complex peptides, and API intermediates, delivered through flexible PO-based (mg → kg) or dedicated FTE models. Complementing these capabilities, our analytical division performs method development, validation, transfer, and stability testing via LC-MS, HPLC, FT-IR, UV–Vis, Karl Fischer, TGA, and more. Each project is supplied with complete data packages (COA, MSDS, NMR, MS, HPLC) and executed under USP, EP, BP, and ICH guidelines—ensuring reproducibility, traceability, and global regulatory acceptance.
At Chemicea, our vision is to be the most trusted global partner in the field of pharmaceutical drug development starting from drug discovery to formulation. We are committed to supporting drug development and regulatory compliance by delivering high-purity, well-characterized reference materials that ensure the quality, safety, and efficacy of medicines.