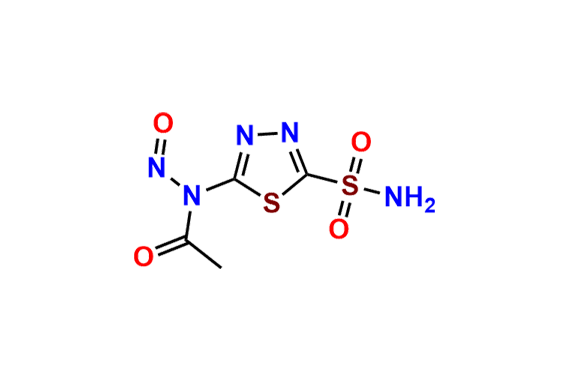
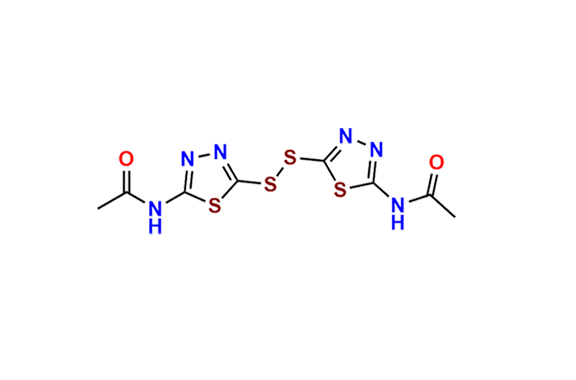
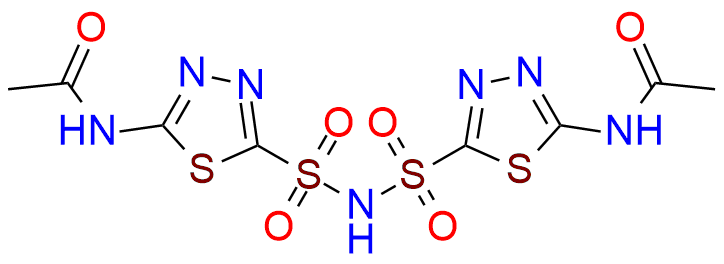
Acetazolamide EP Impurity F
| CAT. No. | CP-A6006 |
|---|---|
| CAS. No. | 80495-47-2 |
| Mol. F. | C8H9N7O6S4 |
| Mol. Wt. | 427.44 |
| Stock Status | In Stock |
- Category: Impurity Standards
- Synonyms: Acetazolamide dimer (USP)
- Chemical Name: N-[5-[(5-Acetamido-1,3,4-thiadiazol-2-yl)sulfonyl]sulfamoyl-1,3,4-thiadiazol-2-yl]acetamide (as per EP)
FAQ
It is a specified impurity of Acetazolamide, formed during synthesis or storage, used in pharmaceutical quality control and regulatory compliance.
It is monitored to ensure quality and safety in Acetazolamide formulations, adhering to guidelines set by regulatory agencies.
It forms as a by-product under specific synthesis or storage conditions, often due to process-related reactions.
Formation can be managed by controlling storage conditions and conducting regular testing to monitor impurity levels.
Detection and quantification of Acetazolamide EP Impurity F employ advanced methods like GC-MS, LC-MS, and HPLC-MS.
Chemicea provides COA, H-NMR, MASS, HPLC, and TGA reports as standard, with additional documents available upon request.
Yes, Chemicea\'s documentation meets standards required by major regulatory bodies like the FDA, EMA, and other authorities.
Acetazolamide EP Impurity F is stable for shipping at room temperature, with specific storage guidelines provided in the COA.
Yes, both standard and customized pack sizes are available based on client requirements.
Chemicea provides Acetazolamide EP Impurity F as a reference standard, supporting impurity analysis, validation, and regulatory compliance.

 VIEW COA
VIEW COA