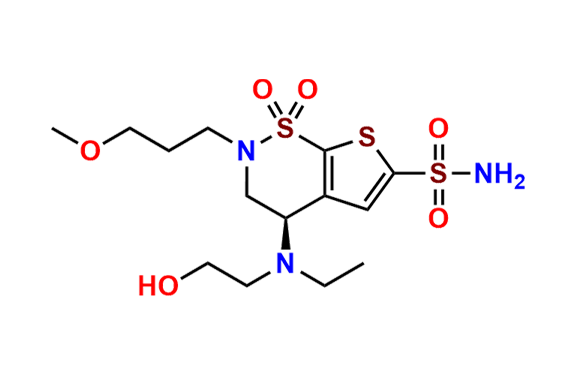
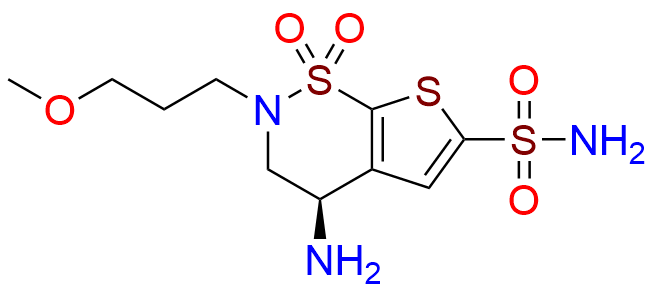
Brinzolamide Impurity D
| CAT. No. | CP-B17004 |
|---|---|
| CAS. No. | 154127-42-1 |
| Mol. F. | C10H16N2O6S3 |
| Mol. Wt. | 356.44 |
| Stock Status | In Stock |
- Category: Impurity Standards
- Synonyms: Brinzolamide (4S)-Hydroxy Analog
- Chemical Name: (S)-3,4-Dihydro-4-hydroxy-2-(3-methoxypropyl)-2H-thieno[3,2-e]-1,2-thiazine-6-sulfonamide 1,1-Dioxide
Request for Quotation : Brinzolamide Impurity D
Search by Keywords - Buy 154127-42-1 | Purchase 154127-42-1 | Order 154127-42-1 | Enquire 154127-42-1 | Price of 154127-42-1 | 154127-42-1 Cost | 154127-42-1 Supplier | 154127-42-1 Distributor | 154127-42-1 Manufacturer | 154127-42-1 Exporter | Brinzolamide Impurity D for Method Validation | Brinzolamide Impurity D for ANDA Filing | Brinzolamide Impurity D for Forced Degradation Studies | Brinzolamide Impurity D Identification Standards | Brinzolamide Impurity D for DMF Filing | Brinzolamide

 VIEW COA
VIEW COA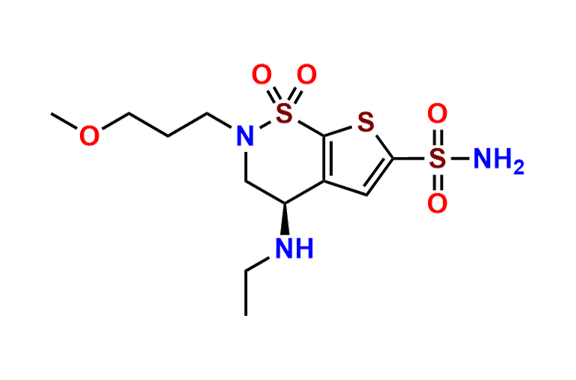


![(R)-4-Hydroxy-2-(3-methoxypropyl)-3,4-dihydro-2H-thieno[3,2-e][1,2]thiazine-6-sulfonamide 1,1-Dioxide](https://chemicea.com/admin/uploads/products/CP-B17009.png)