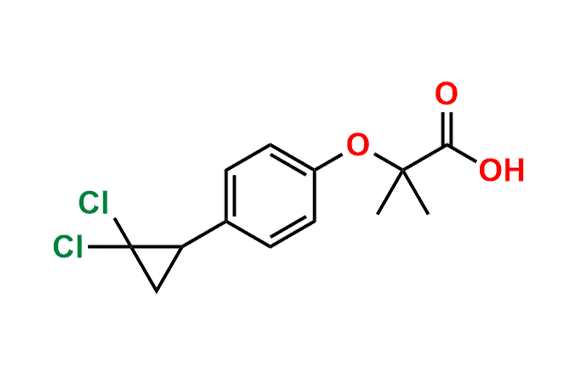
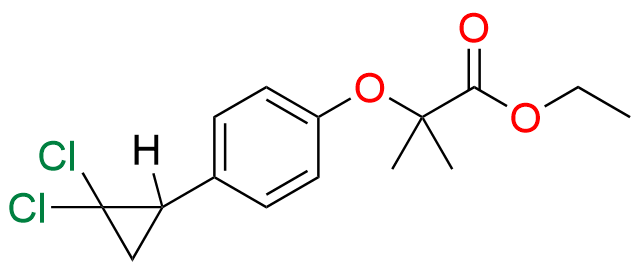
Ciprofibrate EP Impurity E
| CAT. No. | CP-C77005 |
|---|---|
| CAS. No. | 52179-28-9 |
| Mol. F. | C15H18Cl2O3 |
| Mol. Wt. | 317.21 |
| Stock Status | In Stock |
- Category: Impurity Standards
- Synonyms: Ciprofibrate Ethyl Ester
- Chemical Name: Ethyl 2-[4-[(1RS)-2,2-dichlorocyclopropyl]phenoxy]-2-methylpropanoate (as per EP)
Request for Quotation : Ciprofibrate EP Impurity E
Search by Keywords - Buy 52179-28-9 | Purchase 52179-28-9 | Order 52179-28-9 | Enquire 52179-28-9 | Price of 52179-28-9 | 52179-28-9 Cost | 52179-28-9 Supplier | 52179-28-9 Distributor | 52179-28-9 Manufacturer | 52179-28-9 Exporter | Ciprofibrate EP Impurity E for Method Validation | Ciprofibrate EP Impurity E for ANDA Filing | Ciprofibrate EP Impurity E for Forced Degradation Studies | Ciprofibrate EP Impurity E Identification Standards | Ciprofibrate EP Impurity E for DMF Filing | Ciprofibrate

 VIEW COA
VIEW COA