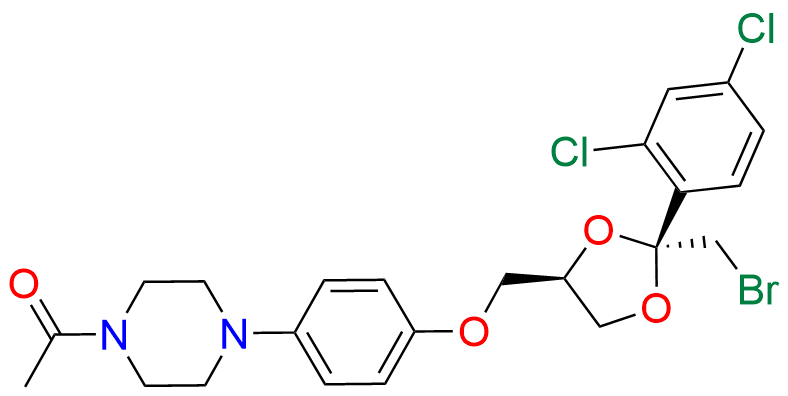
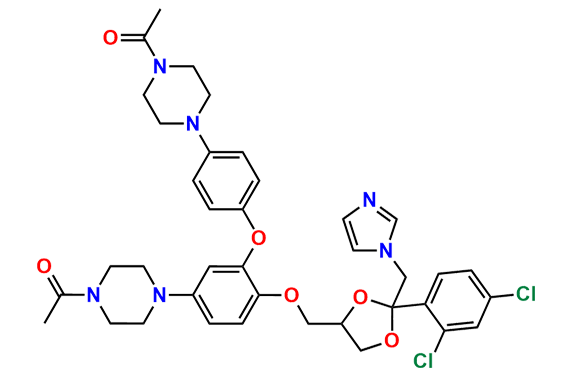
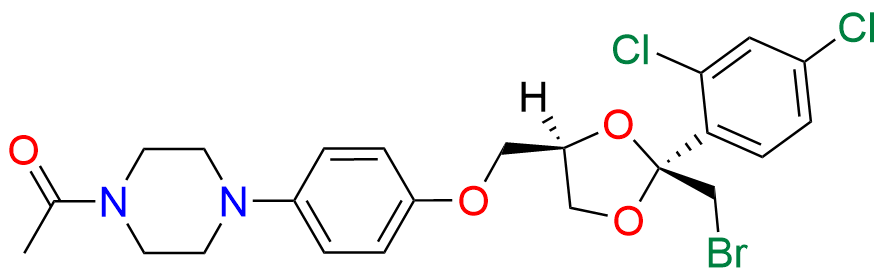
Ketoconazole Impurity 21
| CAT. No. | CP-K2010 |
|---|---|
| CAS. No. | 98207-06-8 |
| Mol. F. | C23H25BrCl2N2O4 |
| Mol. Wt. | 544.27 |
| Stock Status | Custom Synthesis |
- Category: Impurity Standards
- Synonyms: NA
- Chemical Name: 1-(4-(4-(((2R,4R)-2-(bromomethyl)-2-(2,4-dichlorophenyl)-1,3-dioxolan-4-yl)methoxy)phenyl)piperazin-1-yl)ethan-1-one
Request for Quotation : Ketoconazole Impurity 21
Search by Keywords - Buy 98207-06-8 | Purchase 98207-06-8 | Order 98207-06-8 | Enquire 98207-06-8 | Price of 98207-06-8 | 98207-06-8 Cost | 98207-06-8 Supplier | 98207-06-8 Distributor | 98207-06-8 Manufacturer | 98207-06-8 Exporter | Ketoconazole Impurity 21 for Method Validation | Ketoconazole Impurity 21 for ANDA Filing | Ketoconazole Impurity 21 for Forced Degradation Studies | Ketoconazole Impurity 21 Identification Standards | Ketoconazole Impurity 21 for DMF Filing | Ketoconazole