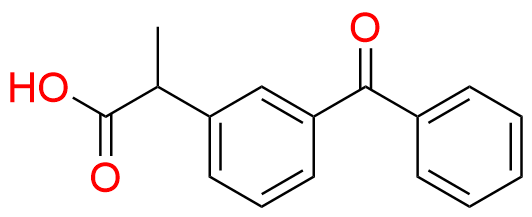
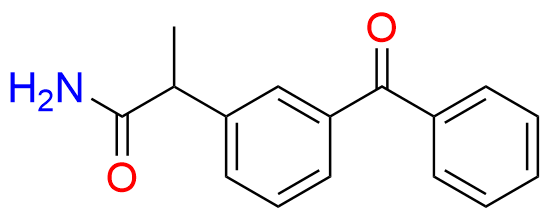
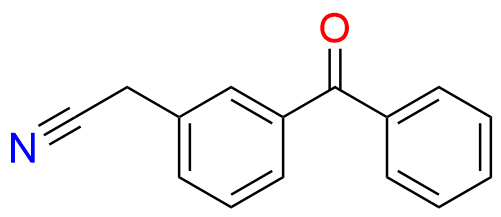
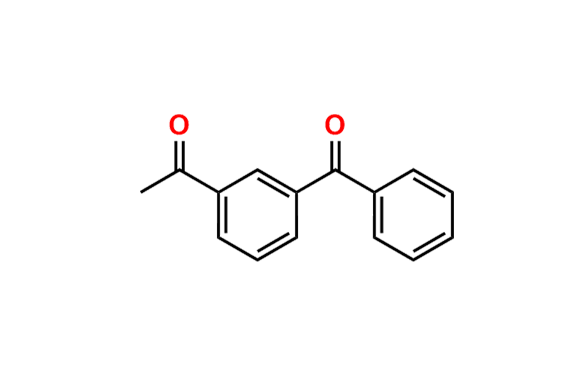
Ketoprofen EP Impurity A
| CAT. No. | CP-K3001 |
|---|---|
| CAS. No. | 66067-44-5 |
| Mol. F. | C15H12O2 |
| Mol. Wt. | 224.26 |
| Stock Status | In Stock |
- Category: Impurity Standards
- Synonyms: Ketoprofen USP Related Compound D
- Chemical Name: 1-(3-Benzoylphenyl)ethanone (as per EP)
FAQ
Ketoprofen EP Impurity A is an impurity that may form in Ketoprofen formulations due to specific synthesis or storage conditions.
Monitoring this impurity is required by regulatory authorities to ensure the safety and efficacy of Ketoprofen formulations.
It forms under specific synthesis or storage conditions that can lead to impurity development in Ketoprofen.
Proper storage and careful control of synthesis conditions can help minimize its formation.
GC-MS, LC-MS, and HPLC-MS are commonly used to detect and quantify Ketoprofen EP Impurity A accurately.
Chemicea provides COA, H-NMR, MASS, HPLC, and TGA reports as standard, with additional reports like CNMR, IR, UV, DEPT, Water content, and CHNS available upon request.
Yes, Chemicea’s documentation meets standards required by major regulatory bodies including USFDA, EMA, ANVISA, and PMDA.
It is stable for shipping at room temperature, with specific storage guidelines provided in the COA.
Yes, Chemicea offers both standard and customized pack sizes to meet specific client needs.
Chemicea supplies Ketoprofen EP Impurity A as a reference standard, aiding regulatory compliance, method validation, and impurity profiling.

 VIEW COA
VIEW COA