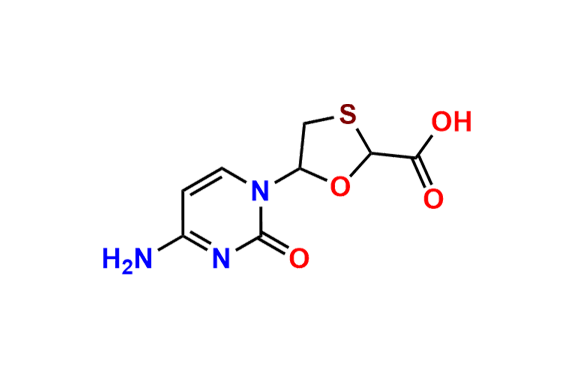
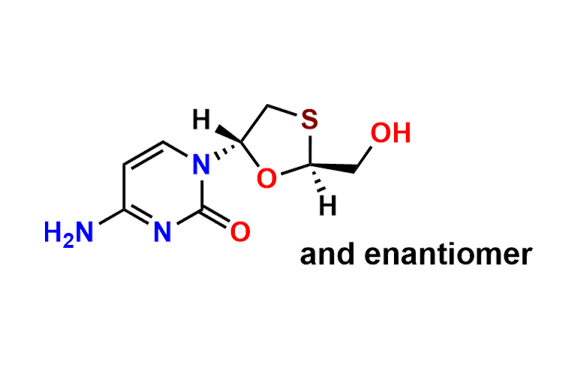
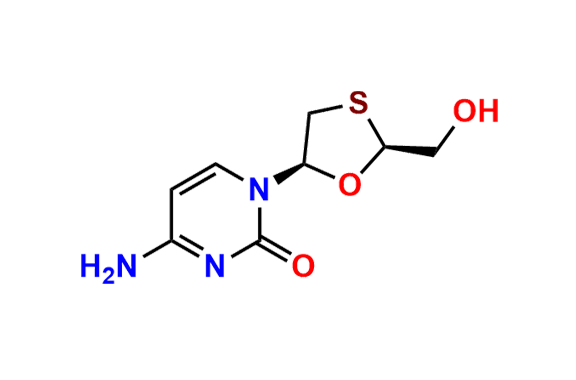
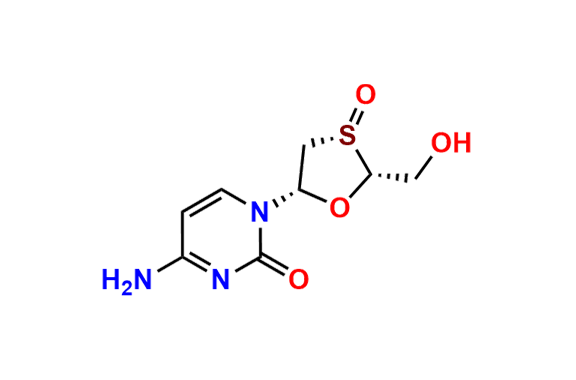
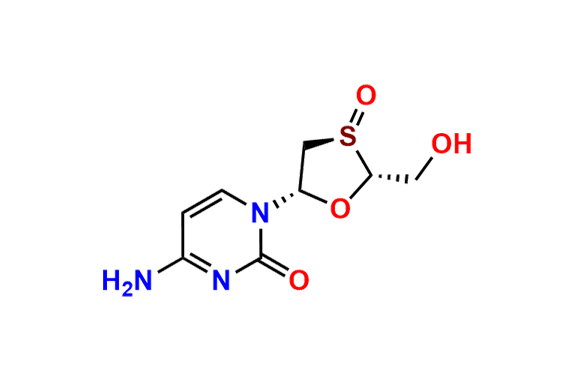
Lamivudine EP Impurity H
| CAT. No. | CP-L3008 |
|---|---|
| CAS. No. | 160552-54-5 |
| Mol. F. | C8H11N3O4S |
| Mol. Wt. | 245.25 |
| Stock Status | In Stock |
- Category: Impurity Standards
- Synonyms: Lamivudine R-sulfoxide
- Chemical Name: 4-Amino-1-[(2R,3R,5S)-2-(hydroxymethyl)-1,3-oxathiolan-5-yl]pyrimidin-2(1H)-one S-oxide (as per EP)
Lamivudine EP Impurity H is a crucial reference standard utilized in the pharmaceutical industry for research and development (R&D). It is widely applied in quality assurance, stability studies, and ensuring compliance with regulatory requirements for Lamivudine-based drug formulations. This impurity is essential for developing analytical methods, conducting toxicological evaluations, and calibrating laboratory instruments for precise and accurate analysis.
Note: This product is strictly for research and development purposes and is not intended for animal or human use.

 VIEW COA
VIEW COA