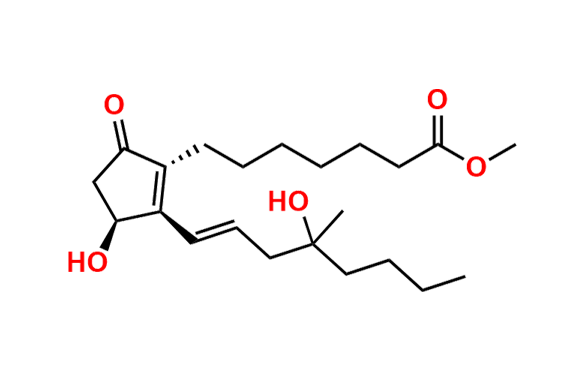
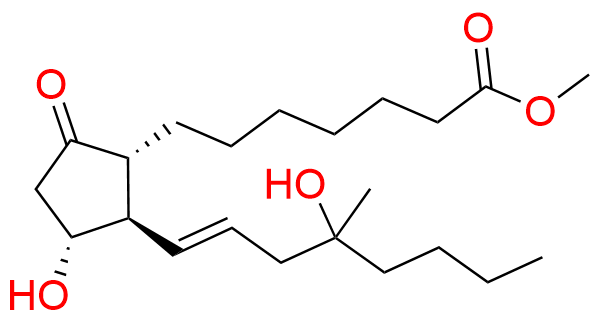
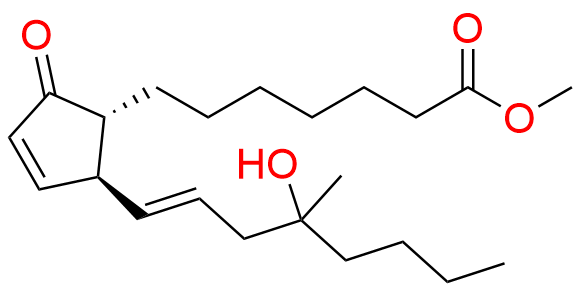
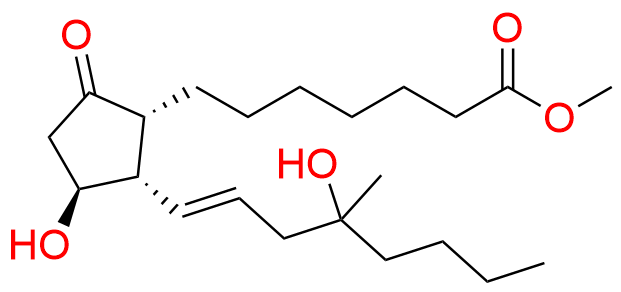
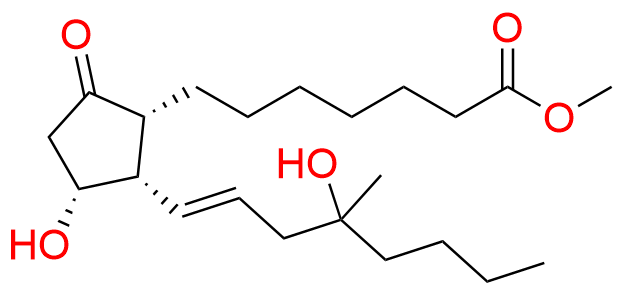
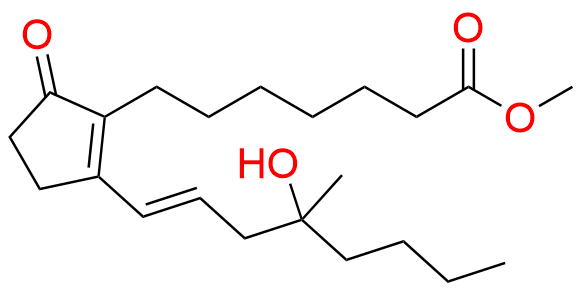
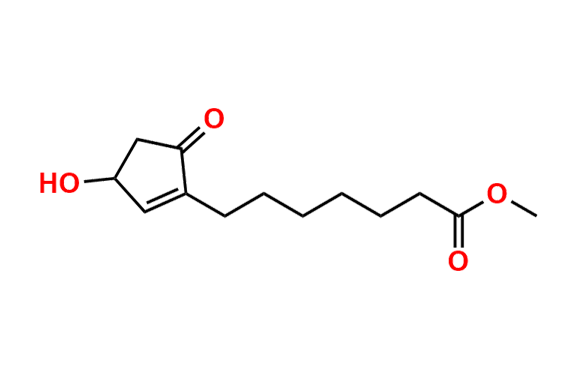
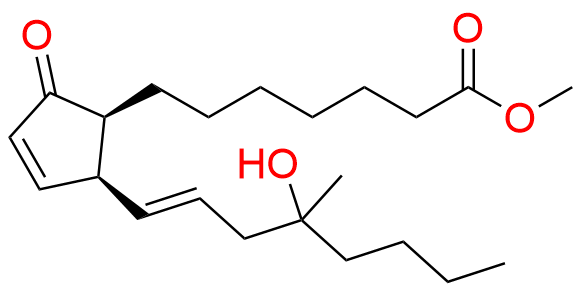
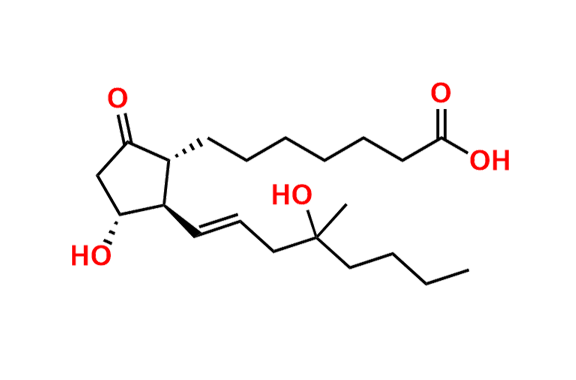
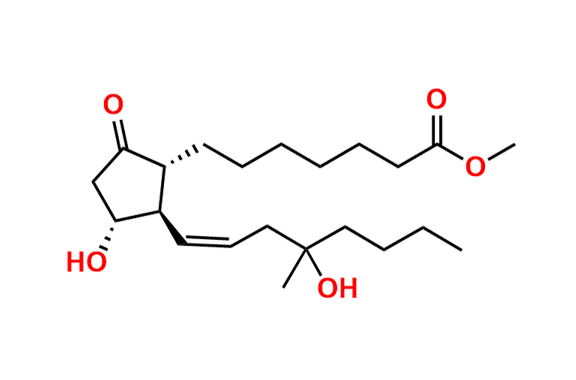
Misoprostol EP Impurity E
| CAT. No. | CP-M22005 |
|---|---|
| CAS. No. | 58717-36-5 |
| Mol. F. | C22H38O5 |
| Mol. Wt. | 382.54 |
| Stock Status | Custom Synthesis |
- Category: Impurity Standards
- Synonyms: 11-epi-Misoprostol
- Chemical Name: Mixture of methyl 7-[(1RS,2RS,3SR)-3-hydroxy-2-[(1E,4RS)-4-hydroxy-4-methyloct-1-enyl]-5oxocyclopentyl]heptanoate and methyl 7-[(1RS,2RS,3SR)-3-hydroxy-2-[(1E,4SR)-4-hydroxy-4-methyloct-1-enyl]-5oxocyclopentyl]heptanoate (as per EP)
Request for Quotation : Misoprostol EP Impurity E
Search by Keywords - Buy 58717-36-5 | Purchase 58717-36-5 | Order 58717-36-5 | Enquire 58717-36-5 | Price of 58717-36-5 | 58717-36-5 Cost | 58717-36-5 Supplier | 58717-36-5 Distributor | 58717-36-5 Manufacturer | 58717-36-5 Exporter | Misoprostol EP Impurity E for Method Validation | Misoprostol EP Impurity E for ANDA Filing | Misoprostol EP Impurity E for Forced Degradation Studies | Misoprostol EP Impurity E Identification Standards | Misoprostol EP Impurity E for DMF Filing | Misoprostol



![Methyl 7-[5-oxo-3(R)-trimethylsilyloxy -1-cyclopenten-1-yl ]heptanoate](https://chemicea.com/admin/uploads/products/CP-M22011.png)
