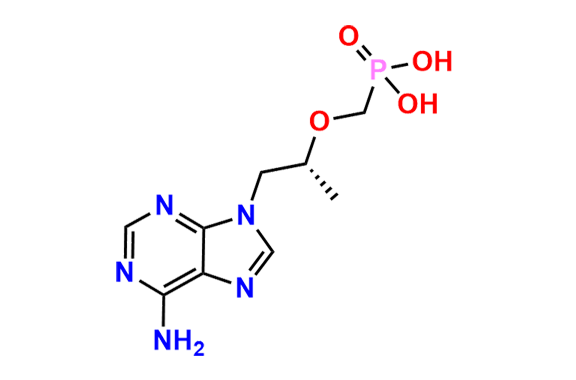
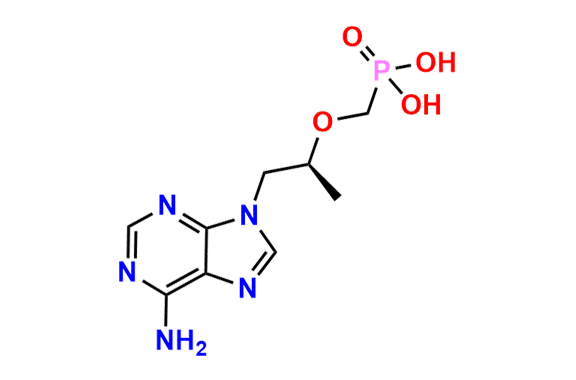
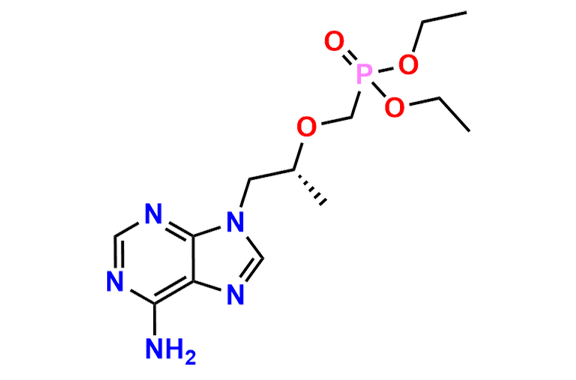
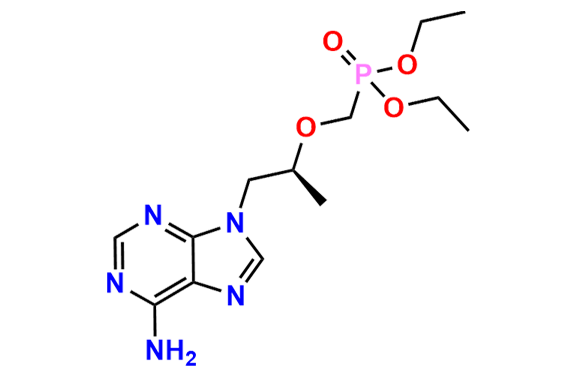
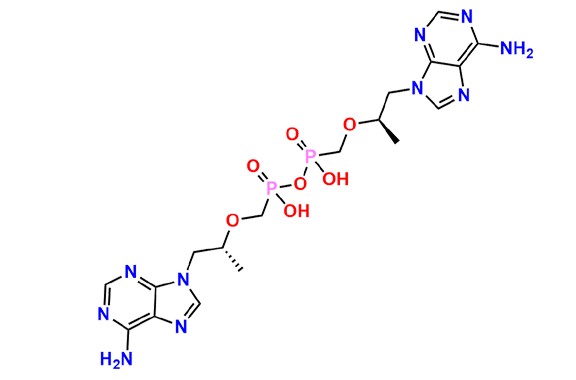
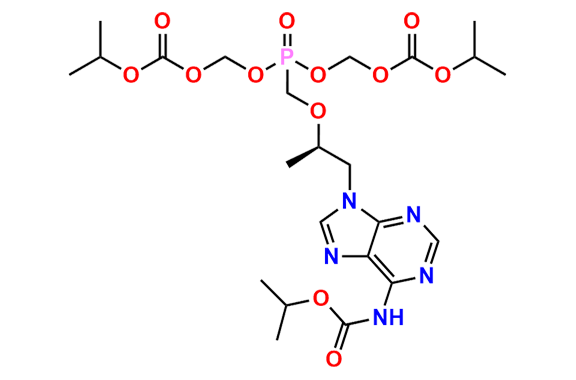
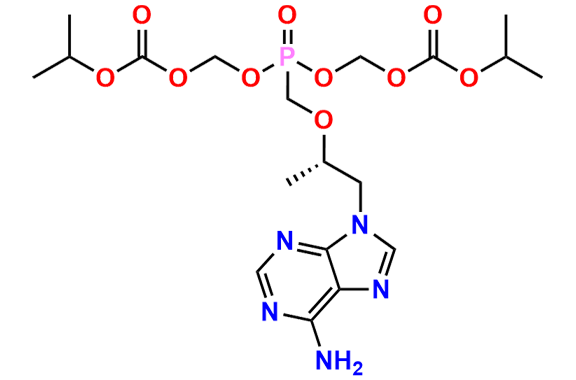
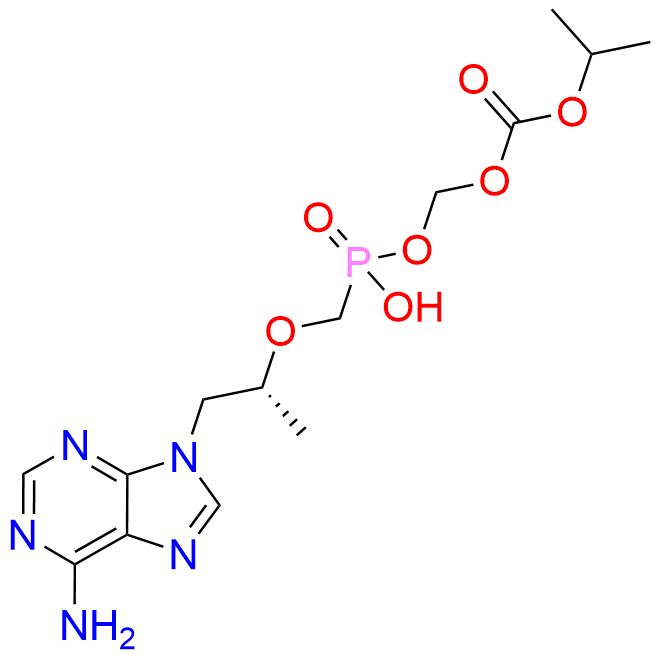
Tenofovir Disoproxil Fumarate IP Impurity D
| CAT. No. | CP-T5001 |
|---|---|
| CAS. No. | 211364-69-1 |
| Mol. F. | C14H22N5O7P |
| Mol. Wt. | 403.33 |
| Stock Status | In Stock |
| Rel. Cas No | 2649753-26-2 (Na salt) ; 1534434-02-0 (Fumarate) |
- Category: Impurity Standards
- Synonyms: Tenofovir Disoproxil IP Impurity A ; Tenofovir Isoproxil Monoester ; Tenofovir Monosoproxil
- Chemical Name: ((((((R)-1-(6-Amino-9H-purin-9-yl)propan-2-yl)oxy)methyl)(hydroxy)phosphoryl)oxy)methyl isopropyl carbonate
Request for Quotation : Tenofovir Disoproxil Fumarate IP Impurity D
Search by Keywords - Buy 211364-69-1 | Purchase 211364-69-1 | Order 211364-69-1 | Enquire 211364-69-1 | Price of 211364-69-1 | 211364-69-1 Cost | 211364-69-1 Supplier | 211364-69-1 Distributor | 211364-69-1 Manufacturer | 211364-69-1 Exporter | Tenofovir Disoproxil Fumarate IP Impurity D for Method Validation | Tenofovir Disoproxil Fumarate IP Impurity D for ANDA Filing | Tenofovir Disoproxil Fumarate IP Impurity D for Forced Degradation Studies | Tenofovir Disoproxil Fumarate IP Impurity D Identification Standards | Tenofovir Disoproxil Fumarate IP Impurity D for DMF Filing | Tenofovir

 VIEW COA
VIEW COA